A Mobile Healthcare Biosensor for Rapid Diagnostic Test: Cancers and Viral Infections
Cancers and viral infections have been the most critical and global health crises in recent decades, especially the recent outbreak of COVID-19. This pandemic has caused the threat of global health, economy, and world-wide frenzy. To interrupt the virus transmission, a rapid, sensitive, and simple diagnostic test is required urgently. Recently, Dr. Hung-Wei Yang, associate professor from the Institute of Medical Science and Technology at National Sun Yat-sen University led an interdisciplinary research team, has successfully developed a mobile healthcare (mHealth) biosensor, a versatile diagnostic tool that can help mitigate the spread of the virus and early detect the risk of cancers to improve the accessibility of global public health care in times like this. Their mHealth biosensor has been proven to accurately evaluate the risk in urothelial carcinomas, prostate cancer and pancreatic cancer. Their research has been published in the high-profile international journals, Biosensors and Bioelectronics and ACS Sensors, in 2020.
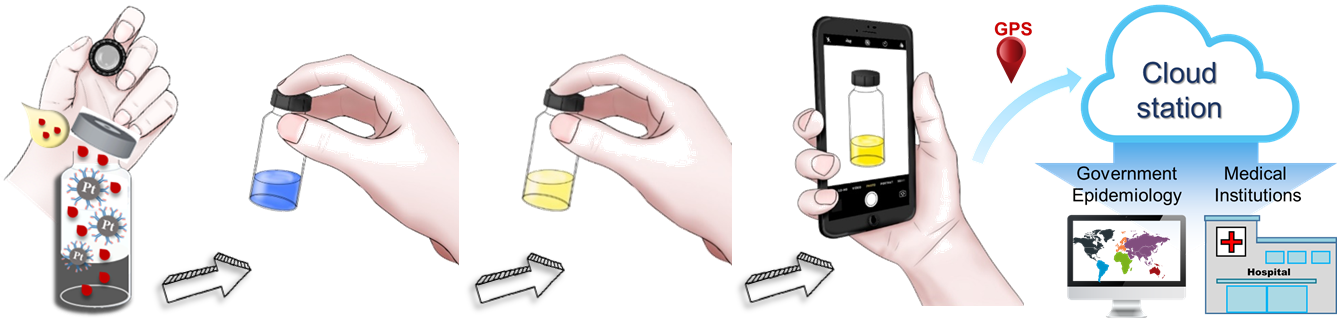
Illustration of the detection procedures for rapid diagnostic test in cancers or viral infections.
Compared to the traditional diagnostic methods, their mHealth biosensor can be performed on whole blood without filtration, and the test can be easily operated by subjects and quantified by a smartphone with 15 min. These characteristics make it ideal for personal use in early cancer diagnosis, postoperative monitoring, and also in ports of arrival and departure for travelers who come from endemic areas as well as for surveillance and geographical tagging for the infected. The mHealth biosensor allows instrument-free and speedy test results in prostate cancer, urothelial carcinoma, lung adenocarcinoma, breast cancer, ovarian cancer, colorectal cancer, Dengue fever, Zika virus, and Ebola virus.
Not long ago, Dr. Hung-Wei Yang’s group also applied this biosensing technology for rapid diagnostic test on COVID-19. It can be used to detect virus spike (S) proteins /or envelope (E) proteins, human immunoglobulin M (IgM) antibodies, and human immunoglobulin G (IgG)] simultaneously within 15 min in human whole blood, serum, or saliva with a limit of detection (LoD) of 0.01 ng/mL, which is much lower than current products (i.e., strip-based rapid diagnostic test with a LoD of 65 ng/mL) for COVID-19 protein-base detection. Through a clinical sample test, their biosensing technology can accurately detect the trace IgM antibodies within 15 min in the blood of the first day of hospitalization of the patient who was tested positive by an RT-PCR nucleic acid analysis, indicating that the developed biosensing technology can be used for rapid diagnostic test of people without obvious symptoms of COVID-19 infection. They expect that this mHealth biosensor will pass clinical trials and come in handy when outbreaks of viral infection diseases happen in the future, and can be further applied to detect communicable diseases, caused by such viruses as Zika, Dengue, and Ebola.

Rapid diagnostic test of IgM/IgG antibodies in whole blood from (A) health people, (B) the patient infected with COVID-19 on the first day after hospitalization, and (C) the patient infected with COVID-19 on the seventh day after hospitalization.
Dr. Yang and his team will continue to work on developing new technologies for mHealth, which will play an active role in combating the spread of viral infection diseases and upgrading the global public health care system.
In the first row, from the left are Postdoctoral Fellow Ying-Tzu Chen, Master student Yi-Jyun Chen, Doctoral students Ying-Pei Hsu and Nan-Si Li. In the back row, from the left are Doctoral student Hao-Han Pang and Assistant of Associate Professor Hung-Wei Yang of the Institute of Medical Science and Technology, NSYSU.




