2019/09 Transmembrane and Coiled-Coil Domain 1 Impairs the AKT Signaling Pathway in Urinary Bladder Urothelial Carcinoma: A Characterization of a Tumor Suppressor
-
Author(s)
Yow-Ling Shiue -
Biography
Prof. Yow-Ling Shiue is currently a Professor/Chair of the Institute of Biomedical Sciences, National Sun Yat-sen University. Her team has recently focused on the biological function and clinical significance of novel membrane proteins in urinary bladder urothelial carcinomas, since membrane proteins are major drug targets.
-
-
Academy/University/Organization
National Sun Yat-sen University -
Source
https://clincancerres.aacrjournals.org/content/23/24/7650.long
-
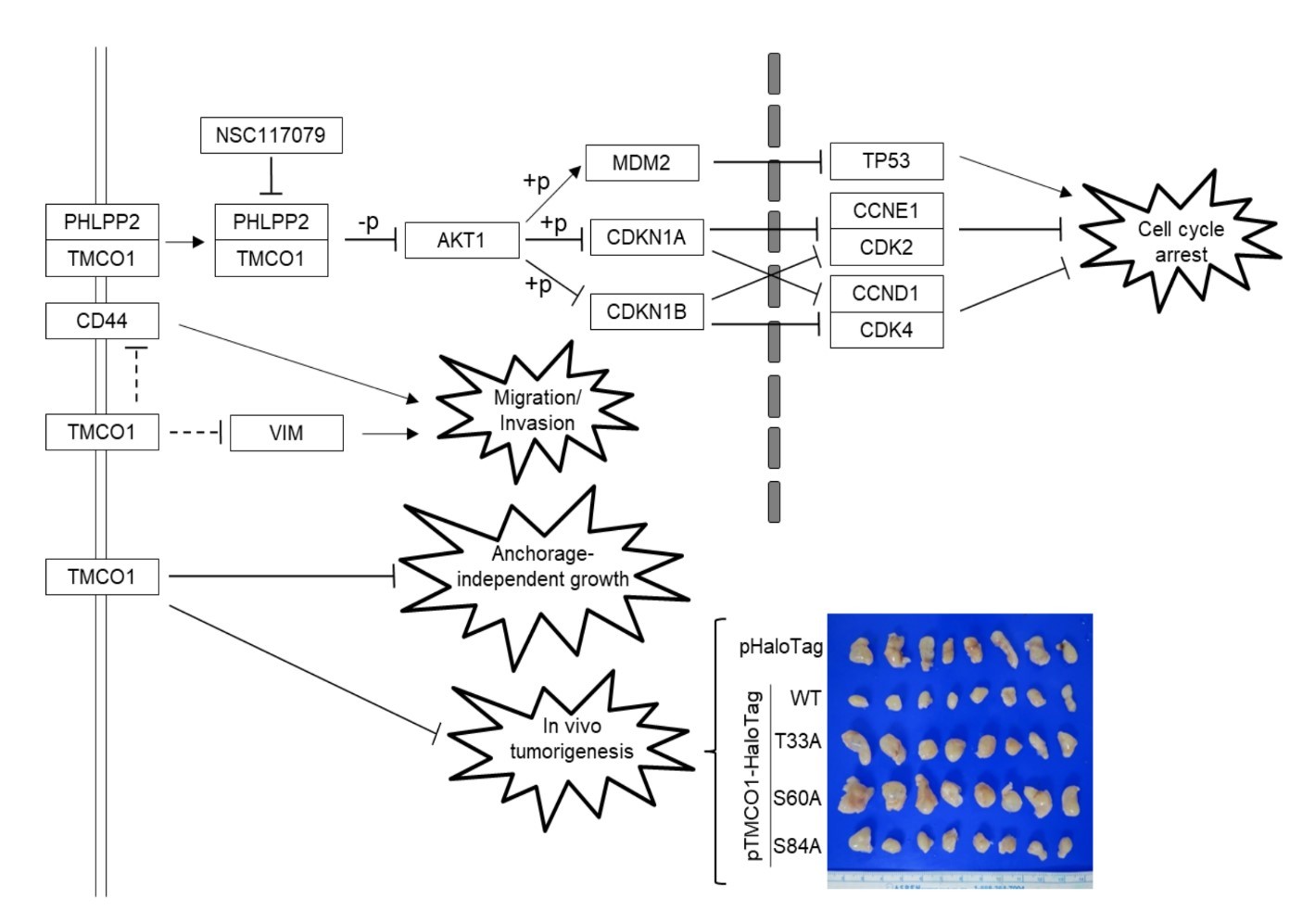
Urinary bladder urothelial carcinoma (UBUC) is a common malignant disease in developed countries. Cell-cycle dysregulation resulting in uncontrolled cell proliferation has been associated with UBUC development. This study aimed to explore the roles of TMCO1 in UBUCs. Data mining, branched DNA assay, immunohistochemistry, xenograft, cell culture, quantitative RT PCR, immunoblotting, stable and transient transfection, lentivirus production and stable knockdown, cell-cycle, cell viability and proliferation, soft-agar, wound-healing, transwell migration and invasion, coimmunoprecipitation, immunocytochemistry, and AKT serine/threonine kinase (AKT) activity assays and site-directed mutagenesis were used to study TMCO1 involvement in vivo and in vitro. Data mining identified that the TMCO1 transcript was downregulated during the progression of UBUCs. In distinct UBUC-derived cell lines, changes in TMCO1 levels altered the cell-cycle distribution, cell viability, cell proliferation, and colony formation, and modulated the AKT pathway. TMCO1 recruited the PH domain and leucine-rich repeat protein phosphatase 2 (PHLPP2) to dephosphorylate pAKT1(serine 473). Mutagenesis at S60 of the TMCO1 protein released TMCO1-induced cell-cycle arrest and restored the AKT pathway in BFTC905 cells. Stable TMCO1 (wild-type) overexpression suppressed, whereas T33A and S60A mutants recovered, tumor size in xenograft mice. Clinical associations, xenograft mice, and in vitro indications provide solid evidence that the TMCO1 gene is a novel tumor suppressor in UBUCs. TMCO1 dysregulates cell-cycle progression via suppression of the AKT pathway, and S60 of the TMCO1 protein is crucial for its tumor-suppressor roles.
.jpg)
Urinary bladder urothelial carcinoma (UBUC) is a common malignant disease, especially in developed countries. Both environmental and genetic factors affect UBUC development. Clinicopathologic features, including histologic grade, stage, size, and multiplicity, are associated with its progression. Despite improvements in surgical techniques and multimodal therapy, 5-year survival rates for patients with muscle-invasive UBUC remain suboptimal. Almost 50% of patients eventually progress and develop systemic disease. Clinical and genetic heterogeneity observed in UBUC patients further complicates the use of general therapies. Cell-cycle dysregulation resulting in uncontrolled cell proliferation has been associated with UBUC development. Thus, targeting a critical molecule for therapies is a rational approach for UBUC treatment. To identify transcripts that are potentially involved in UBUC development, data mining on established expression profiles (GSE31684, n = 93) in the Gene Expression Omnibus database (GEO, NCBI) was performed. One differentially expressed transcript, transmembrane and coiled-coil domain 1 (TMCO1), was identified as being associated with growth factor activity (GO:0008083; molecular function) and was highly expressed in pTa (P = 0.0006) and pT1 (P = 0.0174), compared with pT2-T4 patients with UBUC. The human TMCO1 gene, mapped to human chromosome 1q24.1, encodes a transmembrane protein and is a member of the DUF841 superfamily of several eukaryotic proteins with an unknown function or involvement in any biological process. The 3D structure of TMCO1 protein has not been resolved, yet its topology contains 2 transmembrane domains (#10-30; #91-111) and 1 intramembrane fragment (#138-154). Green fluorescent protein-tagged and Myc-tagged TMCO1 were found to be expressed in the endoplasmic reticulum and/or the Golgi apparatus of COS7 and HeLa cells. TMCO1 mRNA is highly expressed in porcine heart, liver, and kidney. Data mining, and the fact that membrane proteins constitute the largest class of drug targets, prompted us to systematically analyze the relevance of TMCO1 immunoexpression and clinicopathologic features in UBUC patients and its biological significance in vitro and in vivo. In this study, data mining identifies that the TMCO1 transcript is downregulated in the progression of UBUCs. Immunohistochemistry on specimens from 295 UBUC patients showed that TMCO1 downregulation confers poor outcomes in UBUC patients. Changes in TMCO1 level alter cell-cycle distribution, cell viability and proliferation, and colony formation, and modulate the AKT-signaling pathway in vitro. Alterations of the TMCO1 level affect cell migration and invasion in vitro. TMCO1 recruits PHLPP2 to diphosphate pAKT1(S473) in vitro. Mutagenesis on S60 of the TMCO1 protein releases TMCO1-suppressed cell-cycle progression and revises the AKT-MDM2-TP53 signaling pathway.